Temperature has been identified as one of the important variables in cleaning – arguably the most important. So I thought it might be worth some time to develop a little understanding of heat – – especially how it is generated and transmitted.
Heat is a form of energy. The amount of heat contained in an object determines its temperature. The more heat an object contains, the higher its temperature will be. The amount of heat in an object can be increased by adding heat via conduction or radiation from another object at a higher temperature. In the case of conduction, the two objects must be physically in contact with one another while in the case or radiation, heat is radiated as electromagnetic waves from the hotter object and is absorbed by the cooler object. Heat moving from one object to another results in a change of temperature of those objects as heat moves from one to another.
The heat in an object may also be increased by adding work in the form of vibration, bending, etc. In this case, heat is generated by internal friction due to motion.
Note – Heat also moves by convection but that is another process. Convection is the physical mixing of liquids or gasses to achieve a uniform overall temperature.
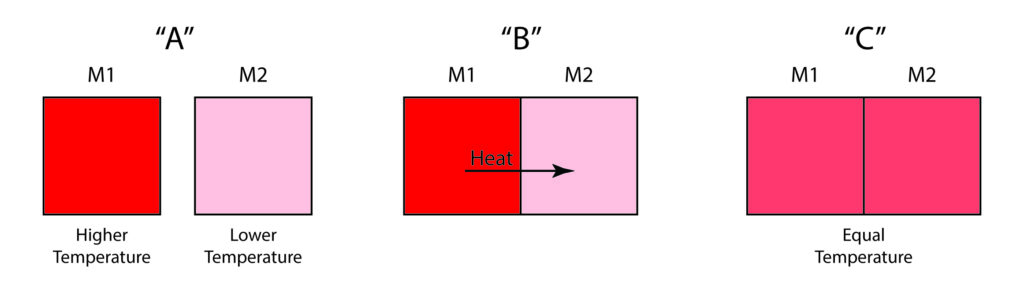
Heat transfer stops when the two objects reach the same temperature. Heat transferred by conduction and/or radiation always moves from the object with the higher temperature to the object with the lower temperature. The speed at which heat moves from one object to another depends on the temperature difference between the two. A greater temperature difference results in a higher rate of heat transfer.
BTU –
Heat is measured in British Thermal Units (BTU’s). The addition of one BTU of heat energy will increase the temperature of one pound of water by one degree Fahrenheit. There are corresponding metric units that apply with calories being the unit of measure for heat and joules being the measure of work.
Specific Heat –
The specific heat of a material is a measure of how much temperature change will be produced by the introduction of a specific amount of heat to a given mass of matter. The specific heat of water is one (one BTU of heat will increase the temperature of a pound of water by one degree Fahrenheit). A lower specific heat indicates that less heat will be required to produce an equivalent temperature change to the same mass while a higher specific heat indicates that more heat will be required.
Heat Capacity –
Different materials are able to absorb different amounts of heat per unit of volume to produce the same temperature change. Specific Heat and Heat Capacity are ratios which are often used interchangeably and are often confused.
Thermal Conductivity –
Thermal conductivity is a measure of the rate of heat transfer across a given thermal gradient over a specific dimension within a material. Higher thermal conductivity indicates faster heat transfer. Thermal conductivity will be discussed in more detail in the next blog.
– FJF –

 Water – De-ionized – Hints
Water – De-ionized – Hints  A Fond Farewell to John Fuchs
A Fond Farewell to John Fuchs  Millipore Testing – Evaluation by Particle Counting
Millipore Testing – Evaluation by Particle Counting  Tape Test for Cleaning Revisited
Tape Test for Cleaning Revisited