“Electropolishing,” (often shortened to E-P) is a term frequently heard in cleaning circles. Electropolishing of surfaces of cleaning equipment is employed to provide enhanced cosmetic appearance and, more importantly, to enhance their functionality.
What is Electropolishing? –
Electropolishing is a lot like electroplating except in reverse. In electropolishing, metal is removed rather than added as it is in a plating process. The equipment is very similar. Parts to be electropolished are immersed in a liquid media called an “electrolyte” while a source of electric current is applied. The part to be electropolished is one electrode called the “anode” and is positively charged. The negatively charged electrode or “cathode” completes the conductive path. The cathode does not enter into the process chemically but its construction can be critical to the success of the process. Other features often include a means to heat the electrolyte and a means of ventilation since gasses evolved during the electropolishing process can be explosive.
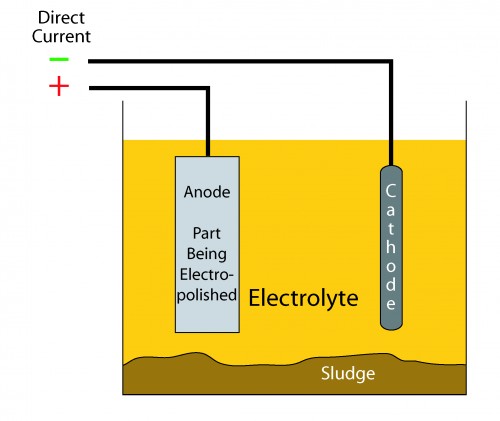
The formulation of the electrolyte is varied depending on the substrate being electropolished and the surface finish desired. Ions removed from the substrate are absorbed into solution in the electrolyte or end up as sludge on the bottom of the processing tank. The electrolyte must be carefully monitored and has a limited useful life.
Prior to electropolishing, surfaces to be electropolished have features undesirable in their intended application. These features may include burrs, slivers or irregularities due to the use of welding, machining or abrasives in the manufacturing process. During electropolishing, features that protrude from the surface act to concentrate current flow (much like a lightning rod attracts lightning) so that they are preferentially attacked by the combination of the electrolyte and the electrical current. The result is that the surface becomes smoother as protrusions are removed.
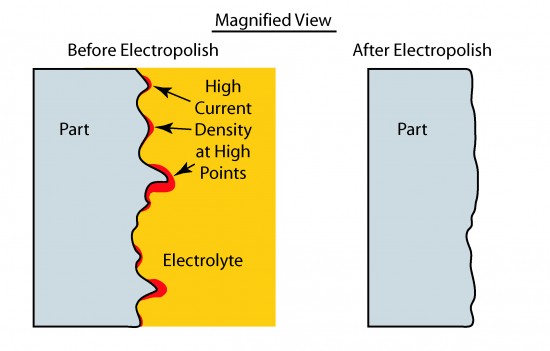
Note – In some cases, the electrolyte used in electropolishing process will dissolve the substrate without the benefit of electric current being applied. However, without electric current, the rate of removal would be more uniform rather than concentrated on the protrusions requiring removal. Adding current helps concentrate the effect to produce the desired result of a smooth surface.
As mentioned above, the cathode, although it doesn’t enter into the process directly can, by its placement and geometry, have a significant effect on the success of the process. Parts with complex geometry may require use of a specially shaped cathode to provide uniform current density as current density may vary depending on the proximity of the cathode and the part being electropolished (anode).
A partial list of things that can affect the electropolishing process are temperature, formulation and freshness of the electrolyte, proximity of the cathode and part (in irregularly shaped parts) and the cleanliness of the part prior to electropolishing. Since, in order to be effective, the electrolyte must uniformly contact the surface being electropolished, any barrier, especially scale, oil, grease and other hydrophobic films must be completely removed prior to electropolishing to prevent a non-uniform result.
Benefits of Electropolishing –
Electropolishing produces a shiny surface nearly mirror-like in appearance. The smooth surface is also easy to clean and prevents the surface from collecting contaminants, especially fine particles and biological contaminants. Another benefit that is somewhat less known is that when stainless steel surfaces are electropolished, nickel and other components of the alloy are preferentially removed leaving a chromium-rich surface with benefits similar to those of passivation and chrome plating.
– FJF –



 English
English Spanish
Spanish Chinese
Chinese Canada
Canada Mexico
Mexico United Kingdom
United Kingdom




One comment on “Electropolishing”
Great article. Very informative information about electropolishing.