In the blog The Effect of Temperature on Ultrasonics, temperature was identified as a major variable in ultrasonic cleaning due to the impact it has on many physical properties of liquids. This blog addresses the effect that chemistry has on the physical properties of liquids and the resulting effect on ultrasonics. Most ultrasonic cleaning utilizes chemistry along with water to enhance either the dissolution (in the case of soluble contaminants) or wetting (in the case of insoluble contaminants) to enhance the cleaning process. The use of plain water is common in rinses although chemistry is occasionally used for purposes of rust prevention or to promote better rinsing or drying.
The addition of chemistry to water changes its physical properties in a number of ways. The blog that discussed temperature effects used a chart to illustrate changes in liquid properties due to increasing temperature. Below, the same chart format has been used to illustrate the changes in physical properties resulting from the addition of cleaning chemistry to plain water.
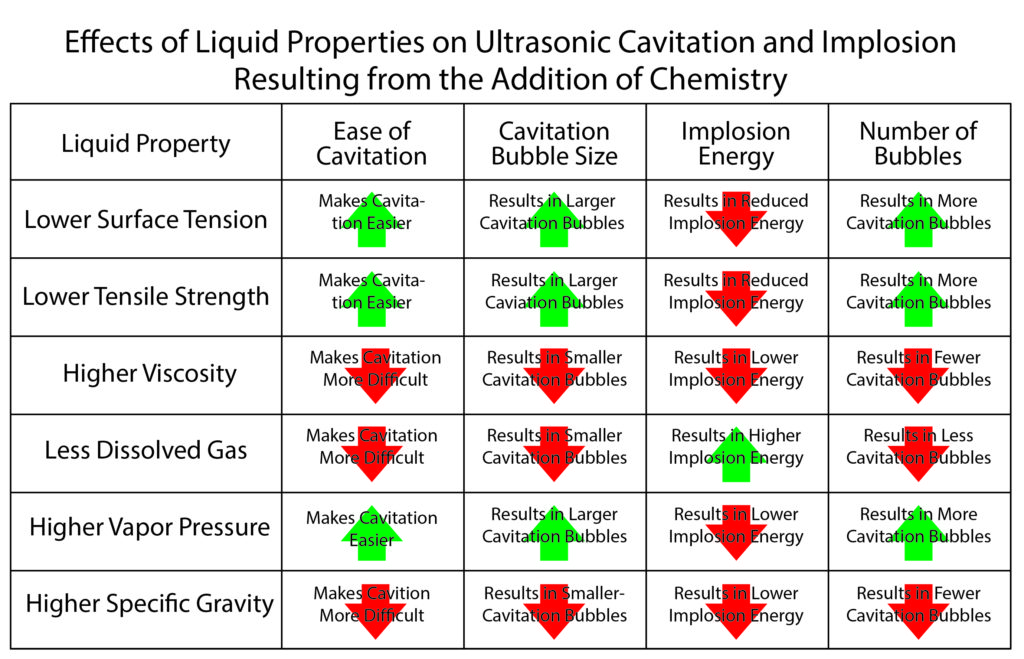
Again, the interactions are complex with one effect of a property change offsetting another in many instances. At first glance, it would appear that the addition of cleaning chemistry has a generally negative effect on the liquid properties favorable to the cavitation and implosion that are essential for ultrasonic cleaning. Based strictly on physical properties this is probably the case. However, the benefit of using chemistry in cleaning offers substantial benefits that outweigh any negative effect. Chemistry is essential to cleaning in most instances. The “take away” here is that anything that can be done in the formulation of chemistry to move the physical properties of the cleaning solution in the direction of favor for cavitation and implosion will further enhance cleaning. We know, for example, that cleaning chemistries that increase vapor pressure (often due to the use of c0-solvents) don’t cavitate well. Emulsions comprised of aqueous and solvent components reduce the tensile strength of the liquid do not cavitate well. This is not obvious in the above chart because, as discussed in the blog related to temperature, although there are more, easily produced and larger cavitation bubbles, their energy is so distributed that they do not implode with sufficient force to substantially benefit cleaning.
Because of these effects, chemistries to be used for ultrasonic cleaning should be tested to verify that they cavitate well and produce high energy implosions. One generalization that can be made is that chemistries intended for spray cleaning seldom support cavitation and implosion. This is due to the fact that for the purposes of spray cleaning, these chemistries are formulated to have relatively low surface tension to prevent the formation of foam which, of course, is not good in a spray washer. The same properties that prevent foaming seem to also prevent effective cavitation implosions in ultrasonic applications.
The benefit of using cleaning chemistry can’t be disputed. Most cleaning chemistries have a favorable range of temperature which should also be observed. Many chemistry formulations have a maximum temperature which, if exceeded, results in separation or “oiling out” (see Chemistry – Tips on Temperature and Concentration) of the chemistry rendering it less or non-effective. In most cases increasing the temperature within the allowable range will enhance the cleaning effect of a chemistry. This, of course, is in addition to the benefits of higher temperature due to physical properties as discussed in the previous post.
– FJF –



 English
English Spanish
Spanish Chinese
Chinese Canada
Canada Mexico
Mexico United Kingdom
United Kingdom




2 comments on “The Effect of Chemistry on Ultrasonics”
Anton – As with nearly any question, the simple answer is, “It depends.” And then the question, “On what?” Well, in this case a bunch of things. First is frequency. The lower the frequency, the higher the amplitude needs to be to achieve cavitation. At very high frequencies in the megasonics range, transducer excursions may only need to be microns or less while at a lower frequency (let’s pick on 25kHz) the requirement may be several thousandths of an inch. The transducer has to move a sufficient distance to produce the size of cavitation bubble that will implode at that frequency. The other things that come into play are temperature, the viscosity of the liquid being cavitated, and all those other factors which have been discussed in the blog. I haven’t heard of many small immersible ultrasonic transducers with application for cleaning. Of course there are medical transducers and sensors that are very small but, remember, in those cases, the object is not cleaning but, rather, just creating a vibration and cavitation is not an issue. -JF
Dear John Fuchs,
Im so happy to have found your blog. May i ask you a simple question?
Id like to build a very small immersible transducer. Is there a minimum Amplitude it needs to have so i can produce cavitations?
Kind regards
Anton